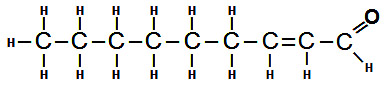When good beer goes bad
Beer is a living beverage that changes over time. From the moment beer leaves the brewery, its flavour characteristics are evolving and changing. Sometimes this is a good thing. Many “bigger” beer styles like Belgian Strong Ales and Imperial Stouts may benefit greatly by being allowed to age in a cellar, like wine, for several months or even years. For beers that are not so robust, the ageing process is often not so kind.
Most styles of beer are at their very best when fresh, and are only negatively impacted by age. As beer ages, it often takes on a stale flavour that may be described as “cardboard”, “paper”, “wet paper”, or “sherry-like”. These flavours will continue to change as a beer ages and may evolve into tastes that can be described as “rotten fruit”, “cooked corn” or even “garbage”.
While sherry-like flavours may not be a bad thing in a Belgian or an Imperial Stout, they are certainly not desirable in less robust beer styles, and the flavours of cardboard, rotten fruit or garbage are never a desired taste in any beer.
The Flavours of Oxidation
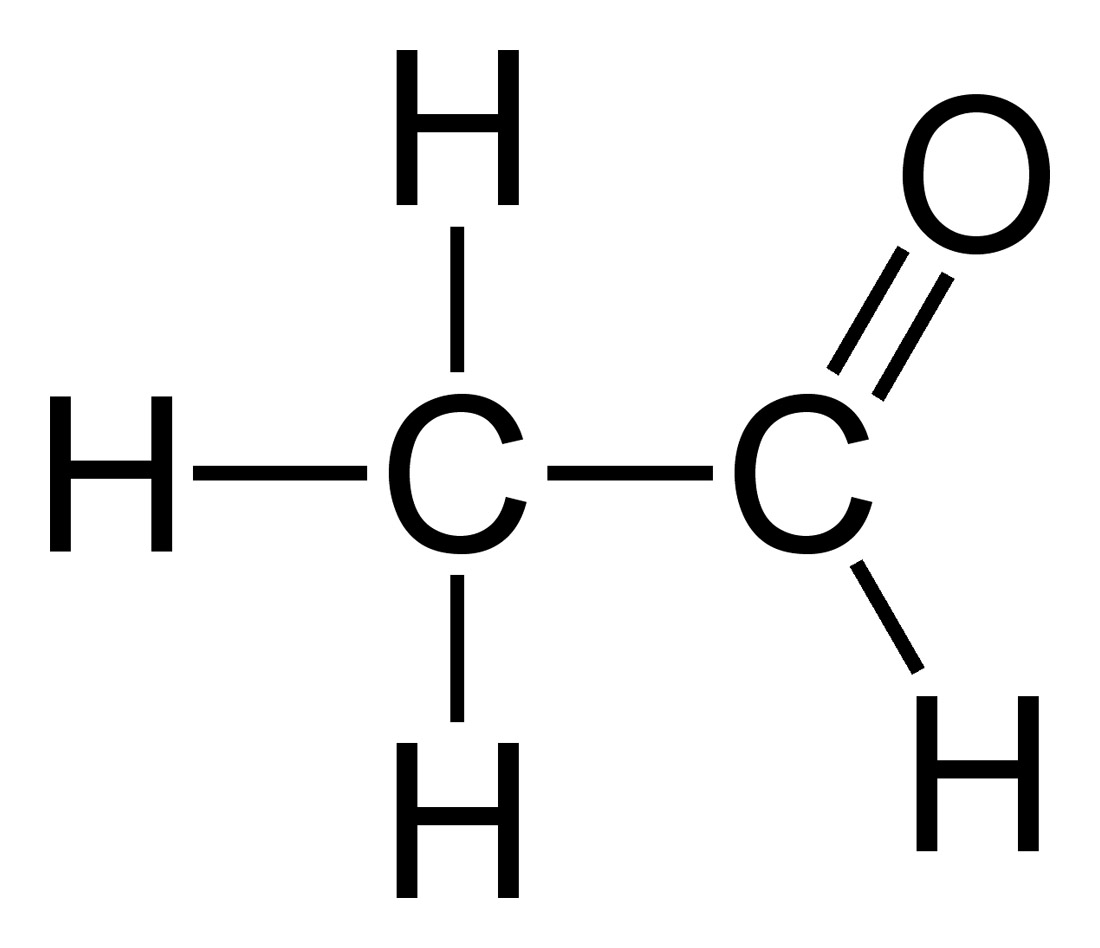
The majority of the stale flavours that develop as beer ages are the result of oxidation. Molecules of the various flavour compounds and alcohols within the beer undergo a chemical reaction with oxygen to form the molecules that are responsible for the stale taste. Fusel alcohols, acetaldehyde and trans-2-nonenal are the primary culprits that are responsible for the majority of the off-flavours associated with stale, oxidized beer, but other compounds also contribute.
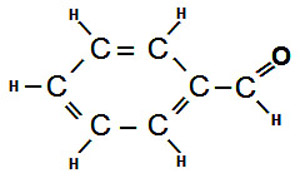
The specific way in which oxidation impacts the flavour and aroma of beer depends on many details that are specific to the particular type of beer that is experiencing oxidation. If trans-2-nonenal is formed in a lighter beer, it may cause a papery or lipstick-like flavour in the beer. Trans-2-nonenal (an aldehyde compound) has a flavour threshold of about 0.1 ppb. The aroma characteristics of many lighter beers can also be affected by oxidation. The malt character that may initially present in the aroma of a fresh beer may change in a way that causes the aroma to be perceived as somewhat “honey-like”. This is due to the formation of 2,3-pentanedione. While this may not necessarily be unpleasant, it is probably not what the brewer originally intended.
Darker, fuller-bodied, beers tend to be affected differently by oxidation. As a darker, fuller-bodied beer becomes oxidized, rich malt flavours and aromas are replaced by sweeter, sherry-like flavours. These sherry-like flavours are the result of the oxidation of the malty-tasting chemicals in beer called melanoidins. Darker, fuller-bodied beers contain lots of melanoidins. The oxidation products of melanoidins are myriad and have a wide range of flavours.
One of the products of the oxidation of melanoidins is benzaldehyde. Benzaldehyde has an almond-like flavour and is a primary contributor to the sherry-like flavour. A small amount of these sherry-like flavours may add complexity to the flavour and aroma of certain strong beer styles like dark Belgian ales and barley wines, but they are not considered appropriate, even in small amounts, in lighter, lower alcohol beers. Even in darker, fuller-bodied beers too much oxidation of the malt melanoidins will eventually cause the original rich malt flavour of the beer to take on a more toffee-like taste, and then, eventually, to be lost altogether.
Another flavour that may be caused by oxidation is the buttery / butterscotch flavour of diacetyl. Diacetyl is formed by the oxidation of alpha acetolactate, a normal by-product of yeast metabolism. Many breweries use brewing techniques to prevent the presence of the alpha acetolactate precursor in the finished beer, but some breweries use techniques that allow this precursor to be present. If alpha acetolactate is present in the finished beer, it will eventually oxidize to diacetyl and the intensity of the characteristic buttery / butterscotch flavour will increase as the beer ages.
The Effect of Heat on Oxidation and Beer Flavour
Oxidation reactions are occurring in beer from the moment the beer is created, but the rate of oxidation, as with most chemical reactions, is accelerated by heat. This means that storing beer cold at all times will help preserve it by reducing the rates of the oxidation reactions associated with the development of off-flavours. Conversely, exposing beer to heat will increase the rates of these oxidation reactions and will increase the rate at which the undesired, off-flavours are developed within the beer.
The temperature inside a non-climate controlled garage may reach 50°C on a hot summer day. The temperature inside a closed car boot (to and from the picnic?) might reach 60°C. If beer is stored at a relatively high temperature for long enough, other off-flavours can develop. At higher temperatures, dimethyl sulfide (DMS) may be produced in beer. DMS has a cooked-vegetable or cooked-corn flavour and aroma.
Another flavour that can develop in beer as a result of prolonged exposure to heat is related directly to the breakdown of yeast. If the beer that has been exposed to high temperatures is bottle conditioned and contains residual yeast, the yeast will begin to autolyze. Yeast autolysis will produce a flavour and aroma that is similar to burning tire rubber.
Freezing Beer
If keeping beer cool helps preserve freshness, then what happens when a beer is frozen? As beer is chilled to a lower and lower temperature, the proteins within the beer have a tendency to precipitate out of solution and form a haze. The proteins begin to denature and their molecular structure is changed. As they denature and form new structures, the proteins may even clump together with other compounds and create small flakes within the beer. Removing proteins from beer changes the texture and mouthfeel of the beer. If a beer is chilled to the point of freezing, the new structures formed by the denatured proteins will quite likely remain as a solid even if the beer is allowed to thaw. Proteins undergo the process of denaturing even at normal storage temperatures and the flakes that develop as a result of this can often be observed in very old beers. The act of chilling a beer to freezing or near-freezing accelerates the formation of the compounds that lead to the development of flakes.
The Effect of Hot / Cold Cycling
Cycling beer temperature from hot to cold can occur during the normal course of the life of a beer consumer. The scenario might go like this.
- Purchase beer from the bottle shop (cold)
- Place beer into the car while doing other shopping (warm-hot)
- Remove beer from car and place it into the refrigerator (cold)
- Remove beer from refrigerator and place into car to take to a mate’s house to watch footy (warm)
- Remove beer from car and place it into the refrigerator at mate’s house (cold)
- Remove leftover beer from refrigerator and place into car for trip home (warm)
- Forget that beer is in the car and leave it there for several days (hot)
The act of cycling beer through hot/cold is not a good thing for the beer. Breweries use freeze/thaw cycles to predict beer shelf-life, but it is largely the “hot” part of the cycle that is doing the damage to the flavour of the beer. Other than denaturing the proteins and formation of flakes in the beer as described previously, extremes of cold are really no bother for the beer.
Conclusions
Oxygen and heat are enemies of beer. Heat increases the rates of the chemical oxidation reactions that are responsible for many of the stale off-flavours that are associated with beer that is past its prime. Beer shelf-life will be longest, and beer quality will be highest if the beer is allowed to remain cold (but not frozen) for the duration of its existence—that is, until you enjoy it.
References:
[adrotate group=”1″]